By their nature, radioactive isotopes are unstable and can change to more stable forms by radioactive decay and emission of different types of radiation. This emitted radiation is useful for cancer treatment which includes placement of “sealed” radioactive sources physically close to the disease site (brachytherapy), or by focusing the radiation emitted by an external source to the disease site (radiotherapy). These are practiced by radiation oncologists. In another clinical specialty of nuclear medicine, radioisotopes are used in a different manner for both diagnostic and therapeutic procedures by intravenous injection of “unsealed” radiopharmaceutical agents. Nuclear medicine allows visualization of the cancer sites by an imaging technique after specifically targeting diagnostic radiopharmaceuticals and subsequently treating them with therapeutic radioactive molecules capable of killing the cancer cells. Combination of such therapy based on evidence derived diagnosis is called theranostics. Lutetium-177 is an important radioisotope used for targeted therapy.
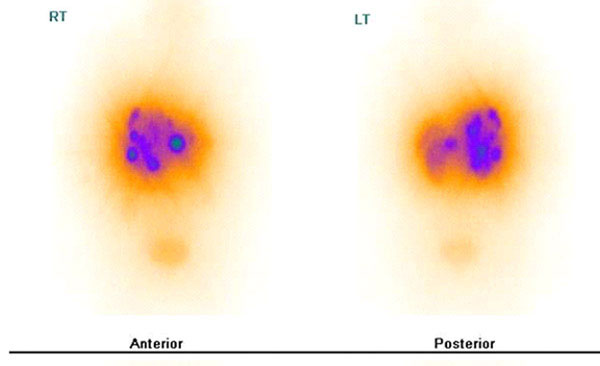
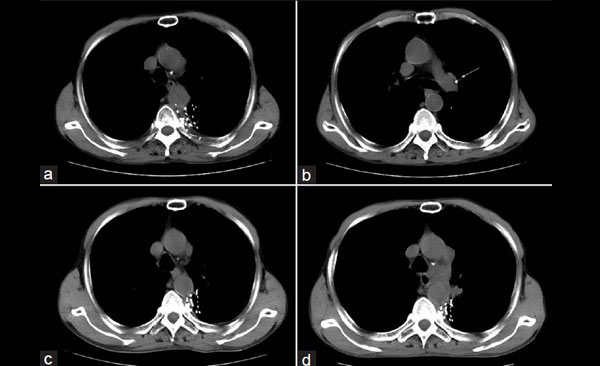
Fig. 1. Single Photon Emission Tomography (SPECT) images of two patients treated with 177Lu agents for cancer therapy. The small dark circular areas represent tumor sites. Images were obtained following the injection and illustrate targeting of tumors for therapy. 177Lu-DOTATATE tumors expressing somatostatin receptors (A, Left) and 177Lu-PMSA agent illustrating well defined prostate metastases (B, Right) (Images courtesy: Ajit Joy, Arun Sasikumar, Raviteja Nanabala, KIMS-DDNMRC, Thiruvanthapuram, Kerala, India).
The strategy for targeted therapy involves stable chemical attachment of 177Lu to molecules which target the disease sites after injection. Radioactive decay of 177Lu emits low energy gamma photons which traverse through tissue and are used for targeting visualization by external imaging. In addition, beta particles are also emitted from 177Lu decay but only travel short distances in tissue, the energy of which is absorbed by the disease target sites killing the cancer cells. The therapy is performed in many parts of the world, as the radioactive levels of 177Lu decrease by only 50% every 6.64 days (half-life) permitting its shipment from the reactor production facility to the clinical sites.
Major clinical applications using 177Lu include treatment of neuroendocrine tumors (NETs) where cellular receptors over-expressed on the surface of the cancer cells are targets for the 177Lu-“DOTATATE” agent, which is an analogue of the hormone somatostatin. The clinical use of this agent is expanding in several countries. Other key examples of over-expressed cancer cell target receptors, many of which are being evaluated for therapeutic targeting with the 177Lu, include bombesin/GRP, CCK/gastrin, endothelin, extendin, integrin, neurotensin, oxytocin and substance P. Another therapeutic example is radioimmunotherpay with 177Lu-labeled rituximab®, a monoclonal antibody useful for treating Non-Hodgkin’s lymphoma. In addition, use of the 177Lu-“EDTMP” agent is progressing in clinical trials for the reduction of metastatic bone pain originating from the spread of cancers from the prostate, ovaries and lungs.
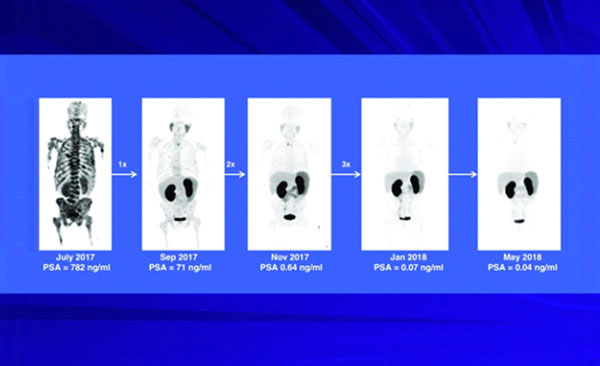
Because of the very high incidence of metastatic prostate cancer throughout the world, a very exciting recent advance uses a 177Lu-based agent for therapy of prostate cancer metastases. The prostate specific membrane antigen (PSMA) protein is expressed in very high levels on prostate parenchyma but in low levels on most other normal cells and is the target for this radiopharmaceutical. PSMA is significantly over-expressed in prostate cancer cells and some other solid tumours. PSMA is an enzyme which hydrolyses the dipeptide, n-acetylaspartylglutamate (NAAG) molecule.
177Lu-NAAG (commonly called 177Lu-PSMA) is emerging as a successful therapeutic agent for metastatic prostate cancer. 177Lu-NAAG binds to PMSA molecules found on the cell surfaces and the complex is then internalized into the target cells providing very high intracellular levels of 177Lu emitting beta radiation for killing the cancer cells. Clinical trials with 177Lu-PSMA are in progress in many countries and have demonstrated effective therapy with very few side effects. The above examples reflect the widespread interest in the clinical community for therapeutic applications of 177Lu- radiopharmaceuticals which are expected to result in regulatory approval and introduction of some of these agents for routine applications.
FF (Russ) Knapp1 and MRA Pillai2
1Emeritus, Medical Radioisotopes Program, Oak Ridge National Laboratory (ORNL), Oak Ridge, TN, USA
2Molecular Group of Companies, Puthuvype, Ernakulam, Kerala, India
Publication
Evolving Important Role of Lutetium-177 for Therapeutic Nuclear Medicine.
Pillai AM, Knapp FF Jr.
Curr Radiopharm. 2015
The information comes from:
https://atlasofscience.org/lutetium-177-radioisotope-targeted-therapy-for-treatment-of-cancer-and-other-diseases/